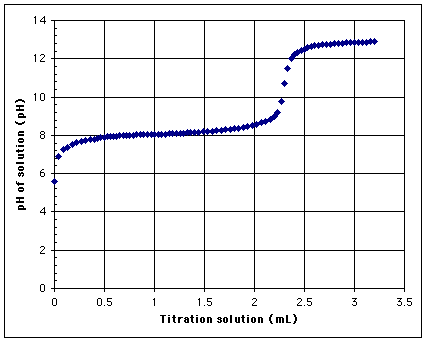
1mL of WVO was mixed with 10mL of isopropyl alcohol. The titration solution was 1g of Red Devil brand lye (NaOH) mixed in 1L of distilled water. This was added one drop at a time to the WVO-alcohol solution. The solution was stirred, and the pH was measured with a Hanna Checker digital pH meter after each drop for 83 drops. The meter was calibrated before the titration. The first drop had a dramatic effect, jumping from 5.6 to 6.9 with one drop. The pH stabilized at approximately 8.0 in the range of 0.7 to 1.2 mL of titration solution. The solution reached a pH of 9 with the addition of 2.2 mL of titration solution. At that point, the pH increased rapidly to a pH greater than 12 where it reached another plateau.
Which value should be considered the appropriate titration level for this case?
a) 0.7 mL where it first reaches pH 8?
b) 1.0 mL in the sweet spot of the plateau?
c) 1.5 mL near the end of the plateau
d) 2.2 mL where the pH reaches 9.0 and starts to increase rapidly?
Of course, this amount would be added to the 3.5 grams of NaOH required for raw vegetable oil. So the range of consideration for this WVO batch is 4.2 grams to 5.7 grams of NaOH per liter of WVO. Comments?